


U.S. Geological Survey, 3215 Marine Street, Boulder; Colorado, U.S.A. (* author for correspondence, e-mail: mmreddy@usgs.gov)
This report was published in the Water, Air, and Soil Pollution 132: 89-104, 2001
Abstract. Negatively charged functional groups of fulvic acid compete with inorganic sulfide ion for mercury ion binding. This competition is evaluated here by using a discrete site-electrostatic model to calculate mercury solution speciation in the presence of fulvic acid. Model calculated species distributions are used to estimate a mercury-fulvic acid apparent binding constant to quantify fulvic acid and sulfide ion competition for dissolved inorganic mercury (Hg(II)) ion binding. Speciation calculations done with PHREEQC, modified to use the estimated mercury-fulvic acid apparent binding constant, suggest that mercury-fulvic acid and mercury-sulfide complex concentrations are equivalent for very low sulfide ion concentrations (about 10-11 M) in Everglades' surface water. Where measurable total sulfide concentration (about 10-7 M or greater) is present in Everglades' surface water, mercury-sulfide complexes should dominate dissolved inorganic mercury solution speciation. In the absence of sulfide ion (for example, in oxygenated Everglades' surface water), fulvic acid binding should dominate Everglades' dissolved inorganic mercury speciation.
Keywords: Florida Everglades, fulvic acid, ion binding, mercury, mercury speciation, PHREEQC, sulfide ion, WHAM
Mercury (Hg) contamination of game fish in South Florida is a major concern for this ecosystem (Hurley et al., 1998; Krabbenhoft et al., 1998; Cleckner et al., 1998). Remediation of Everglades' Hg contamination requires effective management strategies that incorporate understanding of the occurrence, transport and reactivity of dissolved inorganic Hg. For example, Hg interaction with dissolved organic carbon (DOC) and particulate organic carbon (POC) may be important in regulating ecosystem contamination with Hg because of correlation between dissolved Hg and DOC/POC concentrations in natural waters (Driscoll et al., 1994, 1995, 1998; Scherbatskoy et al., 1998; Shanley et al., 1999). Mercury ecosystem contamination is characterized by physical and chemical transformations of Hg- containing species. In particular, Hg solution speciation appears to regulate Hg bioavailability and bioaccumulation. Details of the regulation process are uncertain (Driscoll et al., 1998; Lawson and Mason, 1998; Hurley et al., 1998), however, it is clear that inorganic Hg biogeochemistry influences methylmercury production in aquatic ecosystems (Gilmour et al., 1998). Essential constituents in controlling Hg speciation in natural waters include sulfide species and dissolved organic material (DOM), which includes humic and fulvic acids.
In northern wetlands, Hg bioaccumulation correlates with DOC and/or POC concentrations (Hurley et al., 1994, 1995; Krabbenhoft et al., 1995; Armstrong et al., 1987; Shafer et al., 1997; Bloom et al., 1991; Scherbatskoy et al., 1998; Shanley et al., 1999). Reactivity of DOC is important in South Florida because of high organic carbon production in peat soils and wetlands. However, in the Florida Everglades correlation of Hg bioaccumulation and DOC is absent (Hurley et ai., 1998; Krabbenhoft et al., 1998). Methylmercury concentrations and production rates in the Everglades correlate inversely to sulfide concentration, and insensitivity of Hg bioavailability to DOC has been attributed to formation of Hg-sulfide complexes (Gilmour et al., 1998). Methylmercury, an organic form of Hg, accumulates in aquatic ecosystems and leads to high concentrations of methylmercury in sport fish. Consumption of fish with high methylmercury levels results in increased risk of adverse effects. 'Consumption of contaminated fish is the major source of human exposure to methylmercury in the United States' (Goyer et al., 2000). High-affinity fulvic acid Hg binding sites have been proposed (Allard and Arsenie, 1991). However, there are no definitive measurements of the amount or binding strength of high-affinity sites for Hg in any natural organic material, DOC sample, isolate, or sub-fraction. Objectives of this article are to model fulvic acid interaction with dissolved inorganic Hg ion in the presence and absence of dissolved sulfide ion and to estimate dissolved inorganic Hg species distributions in the Everglades.
A detailed site description has been published (Hurley et al., 1998). Briefly, sampling sites and methodologies used during 1995 evolved to answer specific research questions associated with mercury cycling in the Florida Everglades. Initial sampling in March 1995 focused primarily on the canals and marshes of Water Conservation Area 2A (WCA-2A) and was expanded during summer and winter 1995 (Figure 1).
Water Conservation Area 2A is a 547 km2 constructed marsh in the northern portion of the remnant Everglades. Data used in the modeling reported in this article are from a research study site (site U3) located in WCA-2A (Figure 1). Nearly 60% of the inflow water to WCA-2A is derived from the Everglades Agricultural Area, a 2900 km2 region of farmland (Figure 1). Water input to WCA-2A is from a distribution canal, which receives water from a control structure (connected to the Hillsboro Canal) located along the northern perimeter levee. Flow in the distribution canal is generally slower than in the Hillsboro Canal and is dependent upon the volume delivered through floodgates.
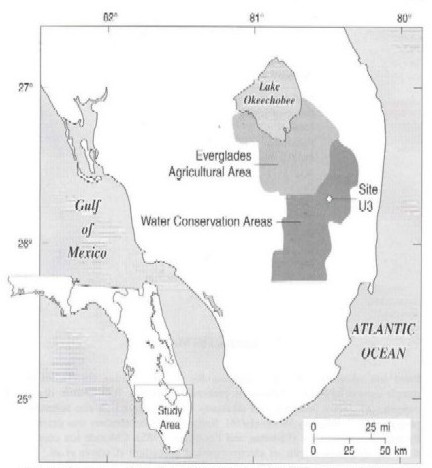
Water discharged to the WCA-2A marsh creates a north-south water quality gradient (Koch and Reddy, 1992). Site U3, in the center of WCA-2A, is less impacted by nutrient-rich canal water and is a reference site for Everglades metal processing.
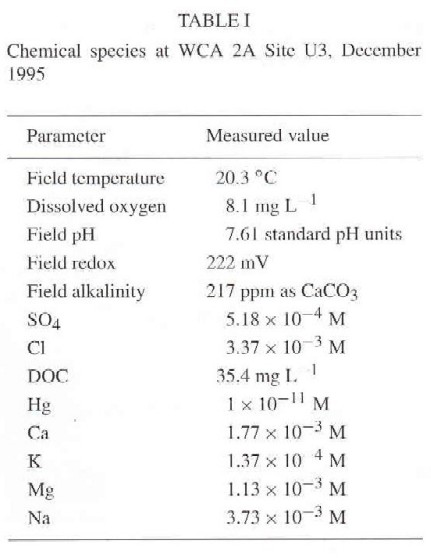
Field and laboratory chemical analyses for a representative Everglades water sample used in model simulations are presented in Table I. Temperature, dissolved oxygen, pH, redox potential and alkalinity were measured on site according to procedures described by Wood (1976). Sulfate ion concentration was determined by ion chromatography (Fishman and Freidman, 1985). Chloride ion concentration was determined with an electrochemical procedure (Cotlove et al., 1958). Dissolved organic carbon measurements were done using a wet oxidation procedure (Aiken, 1995). Major and minor elements were determined using an inductively coupled plasma atomic-emission spectrometric method (Garbarino and Taylor, 1980). Quality control and quality assurance procedures are summarized elsewhere by Reddy et al. (1989).
Dissolved inorganic Hg-fulvic acid interaction and speciation were modeled with a discrete site-electrostatic approach developed by Tipping (1994), which uses techniques pioneered by Marinsky et al. (1999). The model, WHAM (Windermere Humic-Aqueous Model) (Tipping, 1994), is used to describe fulvic acid-Hg ion interaction. Tipping (personal communication, 1995) added Hg-fulvic acid and Hg- humic acid speciation to the version of WHAM used in this report.
WHAM with a simple speciation code characterizes proton binding-site strengths of fulvic acid with a statistical approach (Perdue and Lytle, 1983), and is applicable over a pH range from 3 to 11 and ionic strengths from 0.001 to 1 M. WHAM describes binding of Hg by humic and fulvic substances in terms of ionic complexation at discrete binding sites. Complexation is modified by electrostatic attraction or repulsion, and accounts for nonspecific binding due to counterion accumulation. Mercury speciation in WHAM optimizes the Hg-fulvic acid binding parameters to reproduce published Hg-humic substance speciation data (Kerndorff and Schnitzer, 1980) and thus emphasizes high-abundance Hg binding sites.
PHREEQC, a computer program designed to perform a wide variety of aqueous geochemical calculations, is based on an ion-association aqueous model, and was used to simulate aqueous Hg solution speciation in the presence of sulfide ion (Parkhurst, 1995; Charlton et al., 1997).
Solution redox conditions often control speciation of elements that may exist in several oxidation states. However, redox equilibrium is not established for dissolved Hg and sulfur species in Everglades' surface waters. Non-equilibrium redox conditions are caused by biological (photosynthesis and respiration) and microbiological (sulfate reduction and sulfide oxidation) processes. The coexistence of sulfide and dissolved oxygen, due to the slow oxidation of sulfide in oxygen-containing waters, has been reported (Hoffman, 1977; Millero et al., 1989). PHREEQC simulations assume that Hg redox species are Hg(II). We assume for speciation modeling that dissolved inorganic Hg in solution exists as mercuric ion (Hg+2) and its complexes [Hg(lI)m Ln].
Sulfur redox conditions were defined by the ratio of measured sulfate ion concentration to estimated sulfide concentration. Sulfide species are, therefore, not in redox equilibrium with the oxidizing species present in solution. Dissolved sulfide concentrations are assumed to be at steady state in the PHREEQC calculations. Mercury species in the PHREEQC model are calculated using the MINTEQ data base option. Mercury solution concentrations are estimated based on published data (Hurley et al., 1998).
In the absence of fulvic acid and ligands other than hydroxide ion, Hg speciation simulations demonstrate that the neutral Hg dihydroxy complex [Hg(OH)2] is the predominant solution species in the pH range of interest (pH = 5 to pH = 8).
Solution Hg speciation modeling using WHAM assumes that fulvic acids (about 50% of the DOC) in Everglades' DOC are surrogates for the most important DOC component with respect to Hg binding. Published studies of Hg-humic substance interactions typically employ total dissolved inorganic Hg concentrations that are higher than Everglades' surface-water Hg concentrations. Laboratory measurements at micromolar and greater total dissolved inorganic Hg concentrations favor Hg interactions with high-abundance binding sites such as carboxylate groups present in humic substances (Allard and Arsenie, 1991). If Everglades' organic matter contains a small fraction of very high affinity Hg-fulvic acid binding sites, model predictions based on laboratory studies may not be representative of Hg species distributions in the Everglades (Allard and Arsenie, 1991). However, it is necessary that a successful speciation model describe published Hg-humic substance binding data. Agreement between model simulation results and experimental data is a necessary but not sufficient condition for a speciation model to describe Hg speciation in Everglades' surface waters.
Mercury speciation in the presence of DOC or DOC isolates (i.e., humic and fulvic acids) has been reported for a range of total Hg concentrations. Laboratory studies include: (1) Hg adsorption onto freshly precipitated humic acid at several pH values, metal concentrations and humic acid amounts (Kerndorff and Schnitzer, 1980), (2) quantification of Hg complexation with soil-derived humic substances using an iodide-selective electrode (Yin et al., 1997), (3) use of a gel filtration method to determine the extent of Hg-humic material interaction under conditions of pH and free metal concentration approaching values in natural environments (Mantoura et al., 1978), and (4) fulvic acid-Hg interaction in solutions containing chloride ion using a potentiometric titration procedure (Lovgren and Sjoberg, 1989).
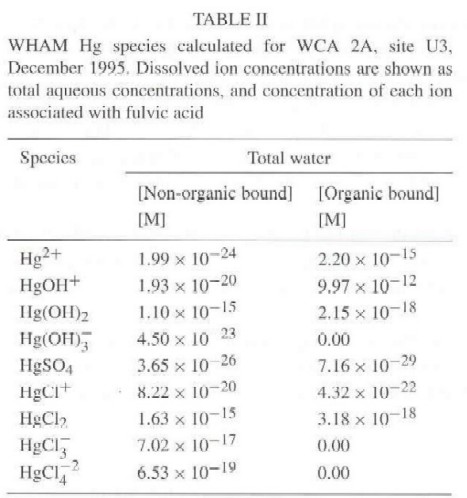
In the presence of fulvic acid, WHAM solution speciation shows that dissolved inorganic Hg exists as a Hg hydroxide-fulvic acid complex (HgOHFA) over a range of solution compositions. This monodentate complex is assumed to form rapidly and reversibly and is the predominant dissolved Hg species above pH = 4, for fulvic acid concentrations typical of the Everglades (Table II). Concentrations in Table II are shown as the Hg-containing ion (non-organic bound, Table II) and the Hg-containing ion bound to fulvic acid (organic bound, Table II). The predominant Hg species (more than 99% of the total dissolved Hg) is the Hg hydroxide-fulvic acid complex (HgOH+-organic bound).
Fulvic acid-Hg binding calculated using WHAM is compared with published laboratory studies by expressing published binding measurements in terms of the fraction of the total dissolved Hg ion bound as fulvic acid complexes (Figure 1). Over the pH range of 2 to 6, Hg-bound fulvic acid complexes least at pH = 2. Non-fulvic acid-bound Hg in solution at pH = 2 exists predominantly as the free mercuric Ion.
At fulvic acid concentrations above 10 mg L-1, the fraction of fulvic acid-bound Hg increases rapidly to 1.0 with increasing solution pH. WHAM simulations employing a fulvic acid concentration of 10 mg L-1 are qualitatively similar to other simulations employing fulvic acid concentrations from 1 to 50 mg L-1. Higher fulvic acid concentrations increase the fraction of Hg bound to fulvic acid at low pH. At 10 mg L-1 the fraction of Hg bound to fulvic acid increased from 0.6 at pH = 2 to 1.0 at pH = 4 and higher.
Mercury speciation is influenced by solution chloride concentration. Increasing chloride ion concentration decreases fulvic acid bound Hg (Figure la) (Lovgren and Sjoberg, 1989). Over the pH range of 3 to 4, inorganic Hg-fulvic acid binding in solutions containing 30-�M Cl- is less than in the absence of chloride ion. Mercury bound to fulvic acid for 30-�M Cl- is 0.4 at pH = 3 and increases to 1.0 at pH = 4. At 120 �M Cl-, the fraction of Hg binding in the presence of varying chloride concentration with changing solution pH. WHAM underestimates the extent of Hg binding at 30 �M Cl- concentration. In contrast, at 120 �M Cl- simulated values agree well with experiments at low pH (3 to 4.5) but differs at pH = 5 (Figure 1a).
Mercury is bound as humic substance complexes at pH = 8 and 0 parts-per-thousand (ppt) salinity in agreement with the WHAM simulations (Figure 1b) (Mantoura et al., 1978). At pH = 8 and 35 ppt salinity the fraction of dissolved Hg bound to fulvic acid is low as is the value calculated with WHAM. Mercury speciation shifts from fulvic acid-bound Hg to Hg-chloride ion complexes, emphasizing the role of Cl- concentration in Hg speciation.
Mercury-humic binding by precipitated humic acid is greater at lower pH than predicted by WHAM at a concentration of 10 mg L-1 fulvic acid (Figure lc) (Kerndorff and Schnitzer, 1980). Model calculations at higher fulvic acid and/or humic acid concentrations agree with the results of Kerndorff and Schnitzer. As the fulvic acid concentration increases in the WHAM simulation, the fraction of fulvic acid-bound Hg increases to 1.0 at lower pH's.
Mercury ion becomes fully bound to humic substance at higher pH (6-7). This result agrees with the results of Yin et al. (1997) - the Hg bound fraction increases with increasing pH (Figure ld). Mercury bound fractions at pH = 4 (0.85 to 0.89), pH = 5(0.92 to 0.96) and pH = 6(0.96 to 0.98) are close to the WHAM prediction (1.0).
Differences in published Hg binding among studies reflect differences in chloride ion concentration. Source material, methods used for isolation and/or purification of humic substances fractions, and/or the experimental procedure used to measure Hg binding also may cause differences in reported bound and free Hg concentrations. In general, the agreement among the studies and WHAM simulations is encouraging. Agreement between model simulations and laboratory results supports use of WHAM for the description of Hg-fulvic acid interactions.
WHAM model simulations predict nine Hg-containing inorganic species (Table II). Currently, WHAM does not include reduced species, such as sulfide ion. WHAM model results were, therefore, used with another computer model (PHREEQC) (Parkhurst, 1995) containing reduced species by estimating an apparent binding constant for Hg hydroxide-fulvic acid (Equation (1)).WHAM simulations show that concentrations of fulvic acid complexes with mercuric ion are several orders of magnitude smaller than the Hg hydroxide-fulvic acid complex. Other dissolved inorganic Hg-containing species are at even lower concentration. Concentration of free Hg-hydroxide complex (HgOH+), Hg hydroxide bound to fulvic acid (HgOHFA-n+1) (Table II) estimated with WHAM and total fulvic acid concentrations in solution (202 �eq L-1), gave an apparent fulvic acid-Hg hydroxide (Equation (1)) binding constant of 3 x 1012 mol L-1.
This apparent Hg-fulvic acid binding constant was added to the PHREEQC (MINTEQ) database (Table III). Species simulations using the apparent humic fulvic acid-Hg binding constants employed a range of sulfide concentrations.
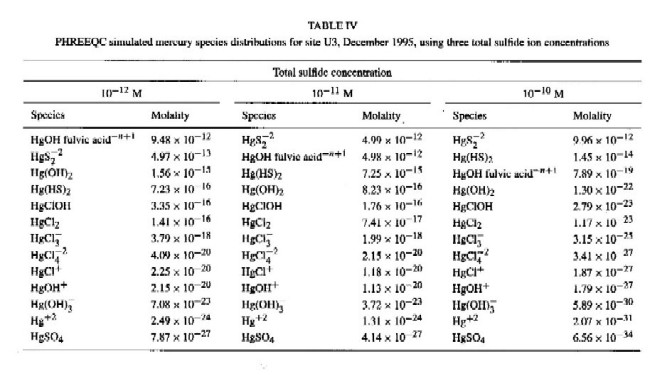
Fulvic acid-sulfide ion competition for Hg ion binding at Everglades' research site U3, in December 1995 (Table IV and Figure 2) illustrates that fulvic acid (35 mg L-1) and sulfide ion bind equal amounts of Hg at a sulfide concentration of 10-11 M with a Hg concentration of 10-11 M. At surface water total sulfide ion concentrations greater than 10-10 M, dissolved inorganic Hg exists primarily as sulfide complexes (HgS2-2 and Hg(HS)02) (Table IV). Surface water sulfide concentrations measured at the WCA-2A site U3 are near or below the measurement detection limit (about 10-7 M). Mercury species distributions estimated with PHREEQC for a range of sulfide concentrations (Table IV) illustrate the importance of sulfide concentration levels in determining Hg speciation in the Everglades' surface water. Where measurable sulfide concentrations (10-7 M or greater) are present in Everglades' surface water, model simulations illustrate that Hg-sulfide complexation will dominate Hg solution speciation. Indeed, model simulations suggest that Hg solution speciation is dominated by sulfide complexes at total sulfide concentrations as low as 10-10 M. Because of high binding strength mercury sulfide predominance in solution speciation is insensitive to the fulvic acid concentration.

Predominance of Hg-sulfide complexes in the presence of sulfide ion is consistent with field data (Gilmour et al., 1998) showing that methylmercury water-column concentrations and methylmercury production rates correlate inversely to solution sulfide concentration. Absence of a DOC influence on Hg methylation in the Everglades is caused by Hg-sulfide complexation at the expense of Hg-fulvic acid complex formation.
In the presence of measurable inorganic sulfide concentrations and low dissolved inorganic Hg concentrations (Table I), PHREEQC Hg species simulations predict that sulfide ion reacts with Hg ionic species in Everglades' surface waters forming Hg-sulfide complexes. However, lack of DOC-Hg interaction in the presence of sulfide ion appears to conflict with studies examining formation and dissolution of mercuric sulfide in the presence of DOC (Ravichandran et al., 1998). Fulvic acid high-affinity Hg binding sites would be expected to be most effective at low dissolved Hg concentrations typical of Everglades' surface water. A detailed analysis of high-affinity fulvic acid Hg binding sites is outside the scope of this article. However, several observations concerning the role of Hg high-affinity binding sites in fulvic acid will be briefly summarized.
Everglades' DOC isolates cause an increase in Hg release from cinnabar (HgS) (Ravichandran et al., 1998). Moreover, hydrophobic (humic and fulvic) acids more effectively dissolved cinnabar than hydrophilic acids and other non-acid fractions of DOC. The presence of various inorganic (e.g., chloride, sulfate, or sulfide) and organic (e.g., salicylic acid, acetic acid, EDTA, or cysteine) ligands, in contrast, does not enhance the dissolution of HgS. These results suggest that Everglades' DOC competes with sulfide for Hg binding causing HgS dissolution and supports the presence of high-affinity Hg binding sites in Everglades' DOC and its fulvic acid fractions.
Laboratory experiments demonstrate that precipitation of metacinnabar (black HgS) is inhibited in the presence of low concentrations of Everglades' DOC isolates (Ravichandran et al., 1999). At low Hg concentrations (less than 5 x 10-8 M), several DOC isolates prevented metacinnabar formation. Metacinnabar precipitation inhibition appears to result from Everglades' DOC-isolate-Hg binding in the presence of high sulfide ion concentrations (about 10-3 M). The effectiveness of Everglades' DOC isolates in enhancing cinnabar dissolution and reducing metacinnabar nucleation suggests that DOC may compete for Hg binding at micromolar sulfide concentrations. Recent studies (Aiken, unpublished) suggest that DOC-isolates influence HgS heterogeneous reactions by adsorption at reaction sites on the mineral surface. For example, prevention of metacinnabar aggregation appears to be due to adsorption of fulvic acid and electrostatic repulsion. The fulvic acid mediation of HgS heterogeneous reaction kinetics may have a mechanism similar to the mechanism of calcite growth inhibition observed with Everglades' DOC isolates (Hoch et al., 2000).
Methylmercury-fulvic acid complex formation cannot be considered in this article because of a lack of published binding studies and appropriate methylmercury- fulvic acid binding parameters for the model WHAM. Limited available results suggest that methylmercury fulvic acid reactions (Hintelmann et al., 1995, 1997) form complexes similar to other types of metal ion organic ligand complexes. Data for methylmercury-fulvic acid binding suggest interactions similar to those of Hg(II)-fulvic acid. As in the case of Hg(II)-fulvic acid interaction (Figure 2) the fraction of methylmercury bound to fulvic acid increases with increasing pH (Hintelmann et al., 1995). Also, as illustrated in Figure 3 for Hg(II)-fulvic acid, (Hintelmann et al., 1997) reports that methylmercury-humic substance complexes predominate in oxidized freshwater with low sulfide concentrations. Methylmercury sulfide complexes are considered by Hintelmann to be the main species present in anoxic waters.
Additional work is needed to characterize the interaction of fulvic acid with methylmercury in the laboratory and in surface and ground waters.
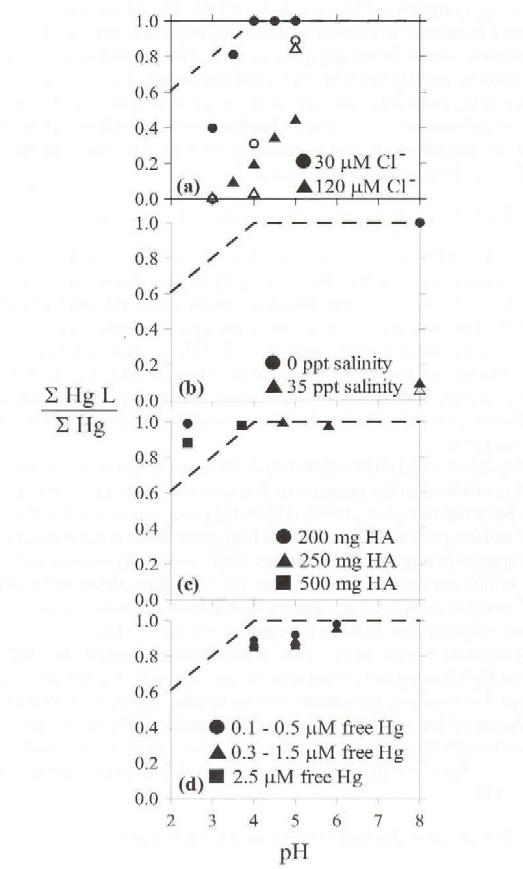
Figure 2. Ratios of the sum of mercury-fulvic acid complexes (Hg(II)L) to the total dissolved inorganic mercury concentration versus pH. The filled symbols represent experimental fulvic acid-bound mercury data from: (a) Lovgren and Sjoberg (1989); (b) Mantoura et al. (1978); (c) Kerndorff and Schnitzer (1980); and (d) Yin et al. (1997). Dashed line represents the WHAM model fit in the absence of chloride ion for one micromolar total dissolved inorganic mercury and 10 mg fulvic acid per liter. Open symbols represent WHAM results at the experimental chloride concentrations.
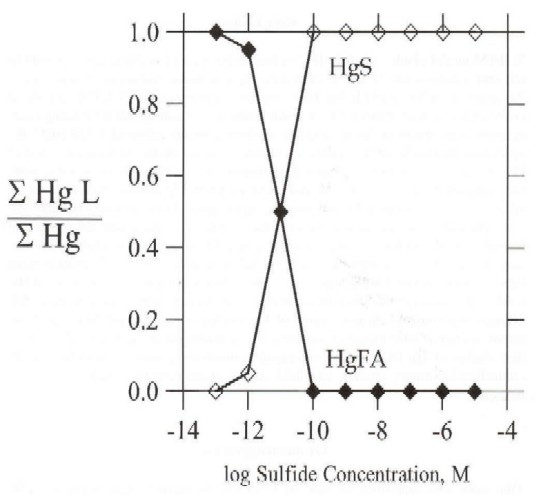
Figure 3. Fraction of fulvic acid- and sulfide-bound inorganic mercury as a function of the total solution sulfide concentration for water composition at site U3, December 1995.
WHAM model results simulate Hg binding to fulvic acid in the absence of sulfide ion and satisfactorily describe published Hg ion-humic substance binding data. An apparent fulvic acid-Hg binding constant estimated from WHAM-calculated concentrations, with PHREEQC, quantifies fulvic acid-sulfide ion Hg binding competition. The apparent fulvic acid-Hg binding constant allowed PHREEQC Hg speciation modeling over a range of sulfide concentrations. Simulations predict Hg-sulfide complexation regulates Hg binding in waters with measurable sulfide concentrations (i.e. 10-7 M and greater). High-Hg-affinity binding sites in fulvic acid may enhance Hg binding by fulvic acid. In oxygenated surface water containing low sulfide ion concentrations, Hg speciation will be dominated by fulvic acid. High Cl- concentrations cause Hg speciation to shift predominantly to Hg chloride complexes from Hg-fulvic acid complexes. Transition from fulvic acid-dominated Hg complexes to Hg chloride complexes may occur at the freshwater-saltwater interface in a coastal environment such as Florida Bay. Additional experimental measurements of Hg binding to humic and fu1vic acid are required over a wide range of solution C1- concentrations and pH values. Further studies of Hg-DOC-sulfide interactions involving computer modeling, well-controlled laboratory studies, and field studies at ambient Hg concentrations are necessary.
This work was supported, in part. by US-EPA Interagency Agreement No. DW 14936802-01-0, the U.S. Geological Survey South Florida Ecosystem Initiative Program and the U.S. Geological Survey Toxic Substances Hydrology Program. We acknowledge the assistance of S. Charlton in modification and implementation of WHAM and PHREEQC and the South Florida Water Management District for assistance and support in field activities. We also thank C. Gunther and P. La Tour for manuscript preparation. Drafting assistance by Micaela B. Reddy is appreciated. We thank reviewers D. Parkhurst, S. Charlton, and L. Fink and two unknown reviewers for comments and suggestions.
Aiken, G. R.: 1995, Environmental Science and Technology 26, 2435.
Allard, B. and Arsenie, I.: 1991, Water, Air and Soil Pollution 56,457.
Armstrong, D. E., Hurley, J. P., Swackhamer, D. L. and Shafer, M. M.: 1987, 'Cycles of Nutrient Elements, Hydrophobic Organic Compounds, and Metals in Crystal Lake', in R. A. Hites and S. J. Eisenreich (eds.), Sources and Fates of Aquatic Pollutants, American Chemical Society, Washington, pp. 491-518.
Bloom, N. S., Walla, C. J. and Hurley, J. P.: 1991, Water, Air and Soil Pollution 56, 477.
Charlton, S. R., Macklin, C. L. and Parkhurst, D. L.: 1997, 'PHREEQCI-A Graphical User Interface for the Geochemical Computer Program PHREEQC', Water-Resources Investigations Report, pp. 1-9.
Cleckner, L. B., Garrison, P. J., Hurley, J. P., Olson, M. L. and Krabbenhoft, D. P.: 1998, Biogeochemistry 40, 347.
Cotlove, E., Trantham, H. V. and Bowman, R. L.: 1958, Journal of Laboratory and Clinical Medicine 51,461.
Driscoll, C. T., Blette, V., Yan, C., Schofield, C. L., Munson, R. and Holsapple, J.: 1995, Water, Air and Soil Pollution 80,499.
Driscoll, C. T., Holsapple, J., Schofield, C. L. and Munson, R.: 1998, Biogeochemistry 40, 137.
Driscoll, C. T., Yan, C., Schofield, C. L., Munson, R. and Holsapple, J.: 1994, Environmental Science and Technology 28, 136A.
Fishman, M. J. and Freidman, L. C.: 1985, 'Methods for determination of inorganic substances in water and fluvial sediments', U.S. Geological Survey Open-File Report, p. 709.
Garbarino, J. R. and Taylor, H. E.: 1980, Applied Spectroscopy 34, 584.
Gilmour, C. C., Riedel, G. S., Ederington, M. C., Bell, J. T., Benoit, J. M., Gill, G. A. and Stordal, M. C.: 1998, Biogeochemistry 40, 327.
Goyer, R. A., Vasken Aposhian, H., Arab, L., Bellinger, D. C., Burbacher, T. M., Burke, T. M., Burke, T. A., Jacobson, J. L., Knobloch, L. M., Ryan, L. M. and Stem, A.: 2000, Toxicological Effects of Methylmercury, National Academy Press, Washington, DC, 290 pp.
Hintelmann, H., Welboum, P. M. and Evans, R. D.: 1995, Water, Air and Soil Pollution 80, 1031.
Hintelmann, H., Welboum, P. M. and Evans, R. D.: 1997, Environmental Science and Technology 31, 489.
Hoch, A. R., Reddy, M. M. and Aiken, G. R.: 2000, Geochimica et Cosmochimica Acta 64, 61.
Hoffman, M. R.: 1977, Environmental Science and Technology 11, 61.
Hurley, J. P., Benoit, J. M., Babiarz, C. L., Schafer, M. M., Andren, A. W., Sullivan, J. R., Hanunond, R. and Webb, D. A.: 1995, Environmental Science and Technology 29, 1867.
Hurley, J. P., Krabbenhoft, D. P., Babiarz, C. L. and Andren, A. W.: 1994, 'Cycling of Mercury Across the Sediment-Water Interface in Seepage Lakes', in L. A. Baker, (ed.), Environmental Chemistry of Lakes and Reservoirs, ACS Advances in Chemistry Series No. 237, American Chemical Society, Washington, pp. 425-449.
Hurley, J. P., Krabbenhoft, D. P., Cleckner, L. B., Olson, M. L., Aiken, G. R. and Rawlik Jr., P. S.: 1998, Biogeochemistry 40, 293.
Kerndorff, H. and Schnitzer, M.: 1980, Geochimica et Cosmochimica Acta 44, 1701.
Koch, M. S. and Reddy, K. R.: 1992, Soil Science Society of America Journal 56, 1492.
Krabbenhoft, D. P., Benoit, J. M., Babiarz, C. L., Hurley, J. P. and Andren, A. W.: 1995, Water, Air and Soil Pollution 80, 425.
Krabbenhoft, D. P., Hurley, J. P., Olson, M. L. and Cleckner, L. B.: 1998, Biogeochemistry 40, 311.
Lawson, N. M. and Mason, R. P.: 1998, Biogeochemistry 40, 235.
Lovgren, L. and Sjoberg, S.: 1989, Water Research 23, 327.
Mantoura, R. F. C., Dickson, A. and Riley, J. P.: 1978, Estuarine and Coastal Marine Science 6, 387.
Marinsky, J. A., Mathuthu, A., Ephraim, J. H. and Reddy, M. M.: 1999, Radiochimica Acta 84, 205.
Millero, F. J., LeFerriere, A., Fernandez, M., Hubinger, S. and Hershey, J. P.: 1989, Environmental Science and Technology 23, 209.
Parkhurst, D. L.: 1995, 'Users guide to PHREEQC - A Computer Program for Speciation, Reaction-Path, Advective-Transport, and Inverse Geochemical Calculations', U.S. Geological Survey Open-File Report, pp. 1-43.
Perdue, E. M. and Lytle, C. R.: 1983, Environmental Science and Technology 17, 654.
Ravichandran, M., Aiken, G. R., Reddy, M. M. and Ryan, J. N.: 1998, Environmental Science and Technology 32, 3305.
Ravichandran, M., Aiken, G. R., Ryan, J. N. and Reddy, M. M.: 1999, Environmental Science and Technology 33, 1418.
Reddy, M. M., Schuster, P. F. and Harte, J. J.: 1989, 'Summary of Data from Onsite and Laboratory Analyses of Precipitation Runoff from Carbonate-Stone Surfaces, National Acid Precipitation Assessment Program, June 1984 to November 1987', U.S. Geological Survey Open-File Report, pp. 1-19.
Scherbatskoy, T., Shanley, J. B. and Keeler, G. J.: 1998, Water, Air and Soil Pollution 105,427.
Shafer, M. M., Overdier, J. T., Hurley, J. P., Armstrong, D. and Webb, D.: 1997, Chemical Geology 136, 71.
Shanley, J. B., Donlon, A. F., Scherbatskoy, T. and Keeler, G. J.: 1999, Water Science and Application 1, 277.
Tipping, E.: 1994, Computers and Geosciences 20, 973.
Wood, W. W.: 1976, 'Guidelines for Collection and Field Analysis of Ground-Water Samples for Selected Unstable Constituents', in W. W. Wood, (ed.), Techniques of Water-Resources Investigations of the United States Geological Survey, Chapter D2, Book 1, Techniques of Water-Resources Investigations of the U.S. Geological Survey, 76-241, pp. 1-24.
Yin. Y.. Allen. H. E.. Huang. CP. and Sanders. P. F.: 1997. Analytica Chimica Acta 341, 73.
