


|
Water chemistry at Snowshoe Mountain, Colorado: mixed processes in a common bedrock A.R. Hoch Lawrence University, Appleton, Wisconsin, U.S.A. M.M. Reddy U.S. Geological Survey, Lakewood, Colorado, U.S.A. The Water Chemistry at Snowshoe Mountain, Colorado: Mixed Processes in a Common Bedrock, 2001, A.R. Hoch and M.M. Reddy, report was publised in the Water-Rock Interaction, Volume 1m edited by Rosa Cidu, Swets & Zeitlinger, Lisse, Pages 533-536. |
|
ABSTRACT: At Snowshoe Mountain the primary bedrock is quite homogeneous, but weathering processes vary as waters moves through the soils, vadose zone and phreatic zone of the subsurface. In the thin soil, physical degradation of tuff facilitates preferential dissolution of potassium ion from glass within the rock matrix, while other silicate minerals remain unaltered. In the vadose zone, in the upper few meters of fractured bedrock, dilute water infiltrates during spring snowmelt and summer storms, leading to preferential dissolution of augite exposed on fracture surfaces. Deeper yet, in the phreatic zone of the fractured bedrock, Pleistocene calcite fracture fillings dissolve, and dioctahedral and trioctahedral clays form as penetrative weathering alters feldspar and pyroxene. Alkalinity is generated and silica concentrations are buffered by mineral alteration reactions. |
|
|
Typically, soil and ground waters with longer residence times in a given rock become more solute rich, but hydrochemical data are often used to interpret solute sources without looking at the reacting minerals. To understand the chemical evolution of water chemistry from dilute snowmelt to spring and stream effluent, we studied water chemistry, hydrology and rock/mineral characteristics in the soil, vadose and phreatic zones developed in an Oligocene welded-tuff near Creede Colorado. Field analyses and experimental results demonstrated the dominance of various solute-producing processes in different hydrologic reaction environments formed from a single bedrock type.
Snowshoe Mountain was originally chosen as a site for a dual watershed study because of the homogeneity of the bedrock (Bates & Henry 1922). It is the eroded central dome of a caldera complex, which was crystallized about 26.5 My before present (Steven & Lipman 1976). High-angle normal faults trend sub-parallel (N-NE) to the Deep Creek fault system in the rock comprised of augite-biotite, quartz latite, welded tuff. This hard, siliceous tuff has been described in detail by Reddy et al. (1994) and Hoch et al. (1999, 2000).
The Seven Parks area is at an elevation of approximately 3500 m and is covered with even-aged, moderate-density Engelmann spruce (Picea engelmanni) and shallow regolith (0.0 - 1.0 m). The mean annual temperature is 2º C, and mean annual precipitation is about 50 cm, about half of which is in the form of snowfall.
Snowshoe Mountain is located in the eastern San Juan Mountains of southwestern Colorado (37º45’ N, 57º30’W). Sampling sites are summarized in Table 1.
Table 1. Sampling sites from highest to lowest elevation.
______________________________________________
Site Elev. (m) Description
______________________________________________
Rock Pit 3505 Zero-tension collector
below 1 m of regolith.
Well 4 (vadose) 3487 13 m well, water level
varies by 10 m.
Well 1 (phreatic) 3475 20 m well, water level
is always near surface.
Site 48 (springs) 3399 Perennial spring, down
gradient from well 1.
Deep Creek gauge 2698 Sampling station at the
base of the watershed.
_____________________________________________
Regolith or "rock pit" water samples were collected and sampled seasonally by zero-tension flow through a collection device described by Claassen et al. (1986).
Wells, springs, and surface waters were sampled seasonally from 1983 to 1998 and analyzed for cations, anions, and silica. (Hoch 1997).
Core was sampled in 3 foot intervals for porosity and permeability tests; thin sections were made from rhodamine dye impregnated billets for optical study of weathering and pore structure (Hoch et al. 2000).
Interstitial glass in soil fragments was analyzed and verified by transmission electron microscopy (Hoch et al. 1999).
Relative dissolution rates of Snowshoe Mountain tuff and augite were determined with a flow-through reactor system (Hoch et al. 1995, 1996).
Mean residence time (MRT) of several points in the shallow aquifer was estimated from seasonal isotope variations in input water (rain and snow) and surface and ground water. The MRT can provide information about ground water recharge and flow paths in the subsurface. Seasonal variations of oxygen-18 in water are attenuated during transit in the shallow subsurface aquifer. This attenuation is related to recharge pathways and the MRT of the shallow subsurface aquifer. This work is in progress.
Rock fragments undergo continual physical disaggregation in the soils. Internal fracturing related to freezing and thawing in soils disaggregates rock fragments with negligible mineral alteration. Phenocrysts such as plagioclase and augite remain unaltered, due to the short duration of wetting by dilute meteoric water (3 to 44 days observed residence time). Interstitial glass is more easily dissolved than reactive phases like biotite and augite (Fig. 1). Dissolution of a residual potassium-rich glass phase (< 5% by volume) produces an anomalous spike of potassium in waters collected from the regolith zone (Hoch et al. 1999).
In Well 4 we observe that water is in contact with the full length of the borehole only 2 to 3 months per year, during and after Spring snow melt. Petrography shows that augite is dissolved in open fractures in core samples from the vadose zone (Fig. 2). We also see preferential dissolution of augite in outcrops where previously subsurface fractures are exposed. Ca/Si and Mg/Si ratios from augite dissolution
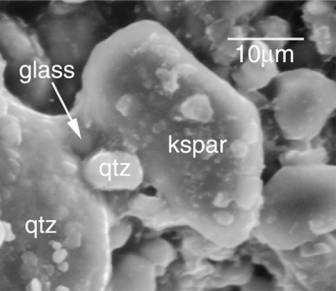
Figure 1. In the soils, interstitial glass is preferentially dissolved and crystalline phases are left unaltered. (qtz = quartz; kspar = potassium feldspar).

Figure 2. Augite is preferentially dissolved in open fractures in core samples from the vadose zone.
experiments mimic ratios in vadose zone waters, supporting the petrographic observation that augite dissolution is the dominant process there (Hoch 1997).
Contrary to other studies that show biotite as a highly reactive phase (Blum et al. 1993), both biotite and hornblende in Snowshoe Mountain tuff are rimmed with relatively inert iron oxide that protects the minerals from weathering. Dissolution of these minerals is not considered to contribute significantly to the chemistry of infiltrating waters.
In fractures near the land surface that contain calcite and a pathway for water, calcite is dissolving but the tuff along the fracture lining remains unaltered, which suggests that the calcite dissolution has only recently begun (Hoch et al. 2000).
In Well 1, water levels remain near the land surface year-round (estimated residence time 741-895 days in the subsurface). In contrast to the vadose well, we observe significant penetrative weathering deeper than the fracture surface (Hoch et al. 2000) and secondary mineralization in core samples.
XRD analysis indicates that both dioctahedral and trioctahedral clays form after feldspar/glass and pyroxene, respectively (Fig. 3). These fill the rock fractures but do not totally impede permeability. (Hoch et al. 2000)
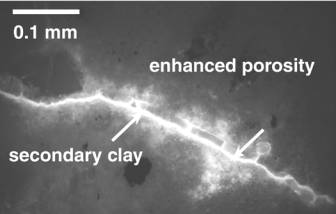
Figure 3. Photomicrograph showing secondary mineralization within an open fracture (long segmented material) and penetrative weathering (bright areas in rock matrix). In samples from the vadose and soil the bulk rock is unaltered.
Potassium is notably enriched in the waters draining the regolith due to dissolution of freshly-exposed glass in the rock matrix (Fig. 4, Hoch et al. 2000). Deeper in the bedrock, fresh surface area production is not a dominant process, interstitial glass is not as available for dissolution and potassium concentrations remain low relative to other dissolved solids. Potassium may also be consumed in the soils by microbial activity.
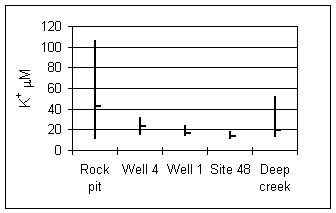
Figure 4. Potassium ion at sampling sites. Vertical bars indicate concentration range, short horizontal bars are means. Data are from Hoch (1997).
Dissolved silica concentrations are constant in ground waters and surface waters, suggesting that they are buffered by secondary formation of silica minerals or clays (Fig. 5). Solid silica phases were detected along with secondary clays in rock fractures, but SiO2 activities do not correspond to any commonly reported minerals (Hoch 1997).
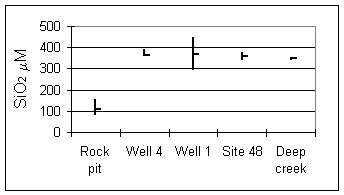
Figure 5. Dissolved silica at sampling sites.
Bicarbonate ion concentrations increase with weathering/residence time (Fig. 6). This results from dissolution of augite along with calcite in fractures where they are present.
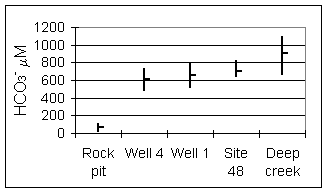
Figure 6. Bicarbonate at sampling
sites.
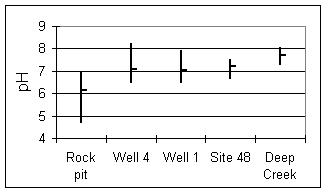
Figure 7. pH at sampling sites.
The pH (Fig. 7) increases only slightly in samples collected deeper than the soil horizon, due to strong buffering by bicarbonate in this pH range.
Calcium ion concentrations (Fig. 8) show little increase in the rock pit (regolith) waters because the main phase dissolving is a potassium-rich glass. At sites with longer residence times, calcium
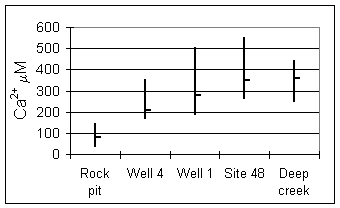
Figure 8. Calcium ion at
sampling sites.
concentrations are greater as a result of augite and calcite dissolution.
For all dissolved constituents shown, well 4 is intermediate between the rock pit and well 1, probably because its waters are a mixture of infiltrating fresh water and deeper, more concentrated, phreatic water.
From the rock pit at the top of the mountain to the stream gauge at the bottom (Table 1), snowmelt input waters are modified in different ways depending on extent of infiltration and available phases for dissolution:
- In the soil or regolith, dissolution of K-rich glass from freshly created rock surfaces is the prevalent chemical weathering process.
- In the vadose zone, chemical weathering occurs on exposed fractures. The most commonly observed weathering process is the preferential dissolution of augite along open fractures, while other minerals, such as plagioclase, biotite and hornblende remain essentially unaltered.
- In cores from the vadose well, calcite was absent, but in those from the phreatic well, calcite is a fracture-filling material and was observed to dissolve near the land surface.
- In the phreatic well, alteration of feldspar, pyroxene, and interstitial glass to clays by penetrative rock weathering was the dominant process, possibly accompanied by reprecipitation of a silica-containing phase that buffers dissolved silica concentrations in springs and creeks.
Although we identified separate and petrographically interesting dissolution processes in the regolith and vadose zone, spring and stream water chemistry is dominated by deeper, slower, penetrative weathering observed in the phreatic zone where residence times are longest.
Bates, C.G., and Henry, A.J. 1922. Streamflow experiment at Wagon Wheel Gap, Colorado. United States Department of Agriculture Weather Bureau, Monthly Weather Review 17: 76 p.
Blum, J.D., Erel, Y., & Brown, K. 1993. 87Sr/86Sr ratios of
Sierra Nevada stream waters: Implications for relative mineral weathering rates. Geochimica et Cosmichimica Acta 58(21/22): 5019-5025.
Claassen, H.C., Reddy, M.M., & D.R. Halm 1986. Use of the chloride ion in determining hydrologic-basin water budgets - a 3 year case study in the San Jaun Mountains, Colorado, U.S.A. Journal of Hydrology 85: 49-71.
Hoch, A.R., Claassen, H.C., Drever, J. I., & M.M. Reddy 1995. Dissolution stoichiometry and near-surface mineral chemistry of augite after 1.5 years in a flow through reactor, with implications for reaction mechanism and watershed-scale mass balance calculations. Geological Society of America annual meeting, New Orleans.
Hoch, A.R., Reddy, M.M., & J.I. Drever 1996. The effect of iron content and dissolved O2 on dissolution rates of clinopyroxene at pH 5.8 and 25ºC: preliminary results. Chemical Geology 132(1-4): 151-156.
Hoch, A.R. 1997. Mechanisms and dissolution kinetics of clinopyroxene and a welded tuff with implications for southwestern Colorado. Ph.D. Dissertation, University of Wyoming, Laramie, Wyoming.
Hoch, A.R., Reddy, M.M., & J.I.Drever 1999. The importance of mechanical disaggregation in chemical weathering in a cold alpine environment, San Juan Mountains, Colorado. Geological Society of America Bulletin 111(2): 304-314.
Hoch, A.R., Reddy, M.M., & M. Heymans 2000. Transient calcite fracture fillings in a welded tuff, Snowshoe Mountain, Colorado. Applied Geochemistry 15(10): 1495-1504.
Reddy M.M., Claassen H.C., Rutherford D.W. & C.T. Chiou 1994. Welded tuff porosity using mercury intrusion, nitrogen and ethylene glycol monoethyl ether sorption and epifluorescence microscopy. Applied Geochemistry 9: 491-499.
Steven, T.A. & P.W Lipman 1976. Calderas of the San Juan volcanic field, southwestern Colorado. United States Geological Survey Professional Paper 958, 35 p.
![[ ]](updated.gif) : Thursday 04 April 02 16:42:43
: Thursday 04 April 02 16:42:43
 .
.