

Biogeochemistry of Carbon and Nitrogen in Aquatic Environments
Research Topics Index Project Home
Nitrate contamination is one of our Nation's most ubiquitous groundwater contamination problems. Project scientists have been using a two-phase approach to develop remediation strategies for nitrate contamination in subsurface water supplies. Both approaches involve using enhanced denitrification as a mechanism for removing nitrate by reducing it to nitrogen gas.
| In the laboratory, we are testing a bench-scale treatment system that uses hydrogen in a bioreactor to enhance denitrification. Hydrogen can also serve as an electron donor for hydrogen-oxidizing denitrifying (HOD) bacteria. These microorganisms are autotrophic, requiring only carbon dioxide as a carbon source, and producing innocuous end-products (nitrogen gas and water) from the nitrate and hydrogen. A patent application has been filed for the hydrogen bioreactor, which has many potential applications. For example, drinking-water suppliers are often faced with high nitrate concentrations in their source waters and the hydrogen bioreactor could be an effective tool for them to supply safe drinking water to their customers. | 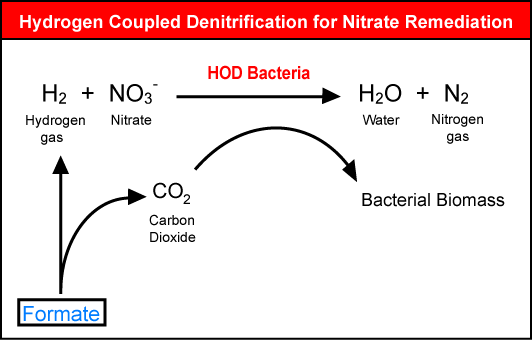
|
|
HOD Bioreactor After 250 Days of Operation
|
 |
Funding: USGS Technology Development Office
USGS Toxic Substances Hydrology Program
Wahlquist, A.M., 2000, The abundance and diversity of autohydrogenotrophic denitrifying bacteria in four
aquifers: MA Thesis, Department of Environmental, Population, and Organismic Biology, University of Colorado,
73 p.
Smith, R.L., Ceazan, M.L., and Brooks, M.H., 1994, Autotrophic, hydrogen-oxidizing denitrifying bacteria in
groundwater, potential agents for bioremediation of nitrate contamination: Applied and Environmental
Microbiology, v. 60, no. 6, p. 1949-1955. (on-line abstract)
|
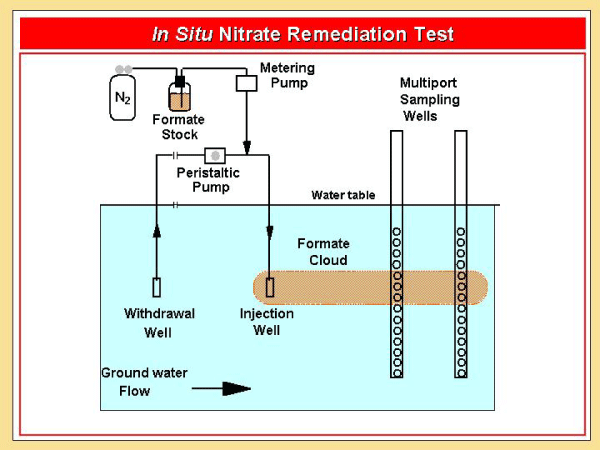
The potential to enhance denitrification in groundwater was investigated by adding sodium formate to the aquifer at the Cape Cod Toxics Substances Hydrology site near Falmouth, Mass. Denitrifying bacteria are native to the subsurface, but are usually electron- donor limited in situ. The addition of formate, which can serve as an electron donor for denitrification, overcomes that limitation, thus, increasing the extent to which nitrate can be consumed. A two- dimensional, numerical transport model was developed for this project to simulate the in situ results. |
|
 |
|
CollaboratorsMark Widdowson, Dept. Civil and Environmental Engineering, Virginia Polytechnical Institute, Blacksburg, Va.
Marc Killingstad, Dept. Civil and Environmental Engineering, Virginia Polytechnical Institute, Blacksburg, Va. |
Field SiteMassachusetts Military Reservation, Cape Cod, Mass. |
Funding
USGS Technology Development Office
USGS Toxic Substances Hydrology Program |
Killingstad, M.W., Widdowson, M.A., and Smith, R.L., 2002, Modeling enhanced in situ denitrification in groundwater:
Journal of Environmental Engineering, v. 128, no. 6, p. 491-504. (on-line abstract)
Smith, R.L., Miller, D.N., Brooks, M.H., Widdowson, M.A. and Killingstad, M.W., 2001, In situ stimulation of
groundwater denitrification with formate to remediate nitrate contamination: Environmental Science and
Technology, v. 35, p. 196-203. (on-line abstract)
| Projects Index Top of Page |
| Return
to Biogeochemistry
of Carbon and Nitrogen in Aquatic Environments Home Page
USGS Home Page | USGS Water Resources | USGS Toxics Hydrology Program | USGS NRP | |