EXAMPLES
The first simulation input data set (table 9) contains four keywords: (1) TITLE is used to specify a description of the simulation to be included in the output file, (2) SOLUTION is used to define the composition of rain water from central Oklahoma, (3) REACTION is used to specify the amount of water, in moles, to be removed from the aqueous phase, and (4) SAVE is used to store the result of the reaction calculation as solution number 2.
TITLE Example 4a.--Rain water evaporation
SOLUTION 1 Precipitation from Central Oklahoma
units mg/L
pH 4.5 # estimated
temp 25.0
Ca .384
Mg .043
Na .141
K .036
Cl .236
C .1 CO2(g) -3.5
S(6) 1.3
N(-3) .208
N(5) .237
REACTION 1
H2O -1.0
52.73 moles
SAVE solution 2
PRINT
-si false
END
TITLE Example 4b.--Factor of 20 more solution
MIX
2 20.
SAVE solution 3
END
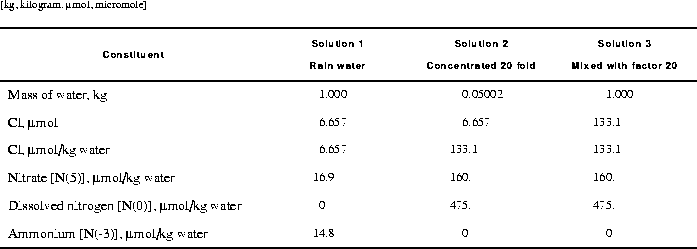
All solutions defined by SOLUTION input are scaled to have exactly 1 kg (approximately 55.5 mol) of water. To concentrate the solution by 20 fold, it is necessary to remove approximately 52.8 mol of water (55.5 x 0.95).The second simulation uses MIX to multiply by 20 the number of moles of all elements in the solution, including hydrogen and oxygen. This procedure effectively increases the total mass (or volume) of the aqueous phase, but maintains the same concentrations. The resulting solution is stored in solution 3 with the SAVE keyword. Solution 3 will have the same concentrations as solution 2 (from the previous simulation) but will have a mass of water of approximately 1 kg.
Selected results of the simulation are presented in table 10. The concentration factor of 20 is reasonable in terms of a water balance for the process of evapotranspiration in central Oklahoma (Parkhurst, Christenson, and Breit, 1993). However, the PHREEQC evaporation modeling assumes that evapotranspiration has no affect on the ion ratios. This assumption has not been verified and may not be correct. After evaporation, the simulated solution composition is still undersaturated with respect to calcite, dolomite, and gypsum. As expected, the mass of water decreases from 1 kg in rain water (solution 1) to approximately 0.05 kg in solution 2 after water was removed by the reaction. In general, the amount of water remaining after the reaction is approximate because water may be consumed or produced by homogeneous hydrolysis reactions, surface complexation reactions, and dissolution and precipitation of pure phases. The number of moles of chloride (mmol) was unaffected by the removal of water; however, the concentration of chloride (mmol/kg water) increased because the amount of water decreased. The mixing simulation increased the mass of water and the number of moles of chloride by a factor of 20. Thus, the number of moles of chloride increased, but the concentration is the same before (solution 2) and after the mixing simulation (solution 3) because of the increased mass of water.
