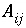 Interfacial area between cells
i
and
j
(m
2
).
Interfacial area between cells
i
and
j
(m
2
).A Temperature dependent constant in the activity coefficient equation.
A 0 Initial surface area of kinetic reacting solid (m 2 ).
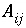 Interfacial area between cells
i
and
j
(m
2
).
Interfacial area between cells
i
and
j
(m
2
).
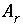 Specific surface area of a surface that is related to a pure phase or kinetic reactant, m
2
/mol.
Specific surface area of a surface that is related to a pure phase or kinetic reactant, m
2
/mol.
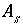 Specific surface area of surface
s
, m
2
/g.
Specific surface area of surface
s
, m
2
/g.
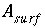 Surface area of a surface-complexation material, m
2
Surface area of a surface-complexation material, m
2
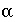 Temperature dependent constant for diffuse layer surface model, 0.02931 [(L/mol)
1/2
C m
-2
] at 25
o
C.
Temperature dependent constant for diffuse layer surface model, 0.02931 [(L/mol)
1/2
C m
-2
] at 25
o
C.
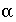 Factor for mobile-immobile exchange, s
-1
.
Factor for mobile-immobile exchange, s
-1
.
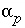 Mole transfer of phase
p
into (positive) or out of (negative) solution, mol.
Mole transfer of phase
p
into (positive) or out of (negative) solution, mol.
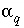 Mixing fraction for aqueous phase
q
.
Mixing fraction for aqueous phase
q
.
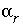 Aqueous transfer of an element between valence states, mol.
Aqueous transfer of an element between valence states, mol.
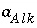 Activity of the master species for alkalinity.
Activity of the master species for alkalinity.
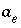 Activity of the master species for exchanger
e
.
Activity of the master species for exchanger
e
.
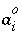 Ion size parameter for aqueous species
i
for extended Debye-Hückel equation or simply a fitted parameter for WATEQ Debye-Hückel equation.
Ion size parameter for aqueous species
i
for extended Debye-Hückel equation or simply a fitted parameter for WATEQ Debye-Hückel equation.
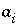 Activity of aqueous species
i
.
Activity of aqueous species
i
.
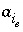 Activity of exchange species
Activity of exchange species 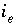 .
.
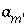 Activity of an aqueous master species, but excluding
Activity of an aqueous master species, but excluding 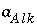 ,
, 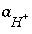 ,
, 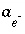 , and
, and 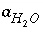 .
.
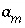 Activity of a master species, including all aqueous, exchange, and surface master species.
Activity of a master species, including all aqueous, exchange, and surface master species.
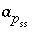 Activity of solid-solution component
Activity of solid-solution component 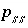 .
.
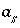 Activity of the master species for surface
s
.
Activity of the master species for surface
s
.
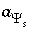 Master unknown for the surface potential of surface
s
,
Master unknown for the surface potential of surface
s
, 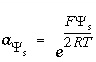 .
.
B Temperature dependent constant in the activity coefficient equation.
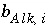 Number of equivalents of alkalinity per mole of aqueous species
i
.
Number of equivalents of alkalinity per mole of aqueous species
i
.
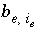 Number of exchange sites of exchanger
e
occupied by exchange species
i
e
.
Number of exchange sites of exchanger
e
occupied by exchange species
i
e
.
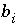 Debye-Hückel fitting parameter for aqueous species
i
.
Debye-Hückel fitting parameter for aqueous species
i
.
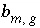 Stoichiometry of element
m
in gas component
g
.
Stoichiometry of element
m
in gas component
g
.
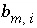 Stoichiometry of element
m
in aqueous species
i
.
Stoichiometry of element
m
in aqueous species
i
.
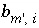 Stoichiometry of element
Stoichiometry of element 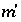 in aqueous species
i
.
in aqueous species
i
.
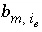 Stoichiometry of element
m
in exchange species
i
e
.
Stoichiometry of element
m
in exchange species
i
e
.
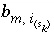 Stoichiometry of element
m
in surface species
Stoichiometry of element
m
in surface species 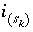 .
.
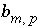 Stoichiometry of element
m
in phase
p
.
Stoichiometry of element
m
in phase
p
.
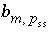 Stoichiometry of element
m
in solid-solution component
Stoichiometry of element
m
in solid-solution component 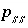 .
.
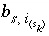 Number of sites of surface type
Number of sites of surface type 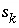 occupied by surface species
occupied by surface species 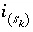 .
.
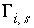 Surface excess of aqueous species
i
for surface
s
, mol m
-2
.
Surface excess of aqueous species
i
for surface
s
, mol m
-2
.
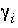 Activity coefficient of aqueous species
i
, kg H
2
O/mol.
Activity coefficient of aqueous species
i
, kg H
2
O/mol.
C Concentration in water, mol/kgw.
C im Concentration in stagnant water, mol/kgw.
C m Concentration in mobile water, mol/kgw.
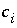 Concentration of aqueous species
i
, mol m
-3
.
Concentration of aqueous species
i
, mol m
-3
.
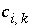 Stoichiometric coefficient of species
i
in the kinetic reaction
k
.
Stoichiometric coefficient of species
i
in the kinetic reaction
k
.
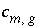 Stoichiometric coefficient of master species
m
in the dissolution reaction for gas component
g
.
Stoichiometric coefficient of master species
m
in the dissolution reaction for gas component
g
.
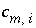 Stoichiometric coefficient of master species
m
in the association reaction for aqueous species
i
.
Stoichiometric coefficient of master species
m
in the association reaction for aqueous species
i
.
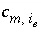 Stoichiometric coefficient of master species
m
in the association reaction for exchange species
i
e
.
Stoichiometric coefficient of master species
m
in the association reaction for exchange species
i
e
.
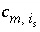 Stoichiometric coefficient of master species
m
in the association reaction for surface species
i
s
.
Stoichiometric coefficient of master species
m
in the association reaction for surface species
i
s
.
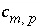 Stoichiometric coefficient of master species
m
in the dissolution reaction for phase
p
.
Stoichiometric coefficient of master species
m
in the dissolution reaction for phase
p
.
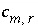 Stoichiometric coefficient of secondary master species
m
in redox reaction
r
.
Stoichiometric coefficient of secondary master species
m
in redox reaction
r
.
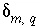 Uncertainty term for the moles of an element or element valence state
m
in solution
q
calculated in inverse modeling, mol.
Uncertainty term for the moles of an element or element valence state
m
in solution
q
calculated in inverse modeling, mol.
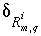 Uncertainty term for the isotopic ratio of isotope
i
for a valence state
m
in the aqueous solution
q
calculated in inverse modeling.
Uncertainty term for the isotopic ratio of isotope
i
for a valence state
m
in the aqueous solution
q
calculated in inverse modeling.
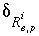 Uncertainty term for the isotopic ratio of isotope
i
for element
e
in the phase
p
, calculated in inverse modeling.
Uncertainty term for the isotopic ratio of isotope
i
for element
e
in the phase
p
, calculated in inverse modeling.
D e Effective diffusion coefficient (m 2 /s).
D L Hydrodynamic dispersion coefficient (m 2 /s).
e Index number for exchangers.
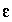 dielectric constant for water, 78.5, unitless.
dielectric constant for water, 78.5, unitless.
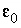 dielectric permittivity of a vacuum, 8.854x10
-12
C V
-1
m
-1
.
dielectric permittivity of a vacuum, 8.854x10
-12
C V
-1
m
-1
.
F Faraday constant, 96,485 Coulomb/mol.
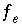 Mole-balance equation for exchanger
e
.
Mole-balance equation for exchanger
e
.
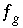 Equation relating aqueous and gas-phase partial pressures for gas component,
g
.
Equation relating aqueous and gas-phase partial pressures for gas component,
g
.
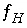 Mole-balance equation for hydrogen.
Mole-balance equation for hydrogen.
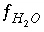 Equation for activity of water in an aqueous solution.
Equation for activity of water in an aqueous solution.
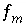 Mole-balance equation for element or element valence state. exchanger, or surface,
m
.
Mole-balance equation for element or element valence state. exchanger, or surface,
m
.
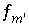 Mole-balance equation for element or element valence state
m
, excluding alkalinity, hydrogen, and oxygen and also excluding the charge balance equation.
Mole-balance equation for element or element valence state
m
, excluding alkalinity, hydrogen, and oxygen and also excluding the charge balance equation.
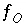 Mole-balance equation for oxygen.
Mole-balance equation for oxygen.
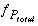 Equation that sums the partial pressures of all gas components, as calculated from aqueous species.
Equation that sums the partial pressures of all gas components, as calculated from aqueous species.
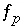 Saturation index equation for phase
p
.
Saturation index equation for phase
p
.
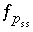 Saturation index equation for component
Saturation index equation for component 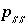 in solid solution
ss
.
in solid solution
ss
.
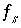 Mole-balance equation for surface
s
.
Mole-balance equation for surface
s
.
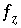 Charge-balance equation for aqueous solution.
Charge-balance equation for aqueous solution.
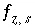 Charge-balance equation for surface
s
, used in explicit diffuse layer calculation.
Charge-balance equation for surface
s
, used in explicit diffuse layer calculation.
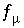 Equation for ionic strength in an aqueous solution.
Equation for ionic strength in an aqueous solution.
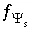 Charge-potential equation for surface
s
, used when diffuse layer composition is not explicitly calculated.
Charge-potential equation for surface
s
, used when diffuse layer composition is not explicitly calculated.
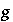 Index number for gas-phase components.
Index number for gas-phase components.
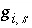 Ratio of concentration of aqueous species
i
in surface excess for surface
s
to concentration in the bulk solution.
Ratio of concentration of aqueous species
i
in surface excess for surface
s
to concentration in the bulk solution.
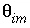 Porosity of the stagnant (immobile) zone (a fraction of total volume, unitless).
Porosity of the stagnant (immobile) zone (a fraction of total volume, unitless).
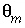 Water filled porosity of the mobile part (a fraction of total volume, unitless).
Water filled porosity of the mobile part (a fraction of total volume, unitless).
i Aqueous species index number.
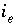 Exchange species index number for exchange site
e
.
Exchange species index number for exchange site
e
.
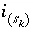 Surface species index number for surface site type
Surface species index number for surface site type 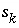 .
.
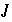 Total number of master unknowns for a calculation.
Total number of master unknowns for a calculation.
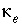 Effective thermal diffusion coefficient (m
2
/s).
Effective thermal diffusion coefficient (m
2
/s).
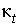 Heat conductivity of the aquifer, including pore water and solid (kJ°C
-1
m
-1
s
-1
).
Heat conductivity of the aquifer, including pore water and solid (kJ°C
-1
m
-1
s
-1
).
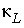 Thermal dispersion coefficient (m
2
/s).
Thermal dispersion coefficient (m
2
/s).
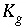 Equilibrium constant for gas component
g
.
Equilibrium constant for gas component
g
.
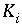 Equilibrium constant for aqueous species
i
.
Equilibrium constant for aqueous species
i
.
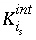 Intrinsic equilibrium constant for association reaction for surface species
i
s
.
Intrinsic equilibrium constant for association reaction for surface species
i
s
.
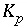 Equilibrium constant for phase
p
.
Equilibrium constant for phase
p
.
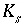 Number of site types for surface
s
.
Number of site types for surface
s
.
k Specific heat (kJ°C -1 kg -1 ); k w for water, k s for solid.
M Total number of master species.
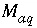 Total number of aqueous master species.
Total number of aqueous master species.
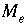 Number of valence states of element
Number of valence states of element 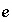 .
.
m Index number for master species.
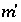 Index number for aqueous master species, excluding
Index number for aqueous master species, excluding 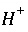 ,
, 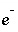 ,
, 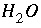 , and the alkalinity master species.
, and the alkalinity master species.
m 0 k Initial moles of kinetic reactant k.
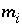 Molality of the aqueous species
i
, mol/kgw.
Molality of the aqueous species
i
, mol/kgw.
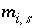 Surface excess of aqueous species
i
, mol/kgw.
Surface excess of aqueous species
i
, mol/kgw.
m k Moles of kinetic reactant k at a given time.
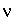 valence of a symmetric electrolyte.
valence of a symmetric electrolyte.
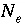 Number of exchange species for exchanger
e
.
Number of exchange species for exchanger
e
.
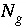 Number of gas components in the gas phase.
Number of gas components in the gas phase.
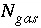 Total moles of gas in the gas phase.
Total moles of gas in the gas phase.
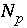 Number of phases in the phase assemblage.
Number of phases in the phase assemblage.
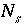 Number of surface species for surface
s
.
Number of surface species for surface
s
.
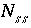 Number of components in solid solution
ss
.
Number of components in solid solution
ss
.
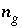 Moles of gas component
g
in the gas phase.
Moles of gas component
g
in the gas phase.
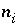 Moles of aqueous species
i
in the system.
Moles of aqueous species
i
in the system.
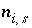 Moles of aqueous species
i
the diffuse layer of surface
s
.
Moles of aqueous species
i
the diffuse layer of surface
s
.
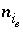 Moles of exchange species
Moles of exchange species 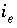 in the system.
in the system.
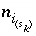 Moles of surface species
Moles of surface species 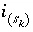 in the system.
in the system.
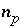 Moles of phase
p
in the phase assemblage.
Moles of phase
p
in the phase assemblage.
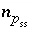 Moles of solid-solution component
Moles of solid-solution component 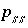 in solid solution
ss
.
in solid solution
ss
.
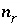 Moles of a reactant, either a pure phase or a kinetically controlled reactant.
Moles of a reactant, either a pure phase or a kinetically controlled reactant.
P Total number of reactants in inverse modeling.
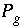 Partial pressure of gas component
g
, atm.
Partial pressure of gas component
g
, atm.
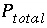 Total pressure in the gas phase, atm.
Total pressure in the gas phase, atm.
p Index number for phases in phase assemblage.
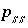 Index number for components in solid solution
ss
.
Index number for components in solid solution
ss
.
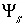 Surface potential for surface
s
, V.
Surface potential for surface
s
, V.
Q Number of aqueous solutions.
q Index number for an aqueous solution in a set of aqueous solutions.
q Concentration in the solid phase, expressed as mol/kgw in the pores.
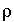 Density (kg/m
3
);
Density (kg/m
3
); 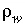 for water,
for water, 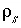 for solid.
for solid.
R Gas constant, kJ mol -1 K -1 .
R Retardation factor (unitless).
R T Temperature retardation factor (unitless).
R Total number of aqueous redox reactions in inverse modeling.
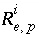 Isotopic ratio of isotope
Isotopic ratio of isotope 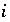 for element
for element 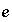 in phase
in phase 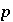 .
.
R im Retardation in the stagnant zone, unitless.
R k Overall rate of reaction for substance k , mol/kgw/s.
R m Retardation in the mobile zone, unitless.
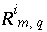 Isotopic ratio of isotope
Isotopic ratio of isotope 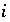 for valence state
for valence state 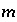 in aqueous solution
in aqueous solution 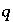 .
.
r k Specific rate of reaction for solid k , mol/m 2 /s.
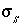 Surface-charge density for surface
s
, C/m
2
.
Surface-charge density for surface
s
, C/m
2
.
s i Concentration of i in the solid phase, mol/kg solid.
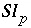 Saturation index for phase
p
.
Saturation index for phase
p
.
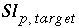 Specified target saturation index for phase
p
.
Specified target saturation index for phase
p
.
SS Number of solid solutions in solid-solution assemblage.
ss Index number for solid solutions.
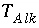 Total number of equivalents of alkalinity in solution.
Total number of equivalents of alkalinity in solution.
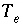 Total number of equivalents of exchange sites for exchanger
e
.
Total number of equivalents of exchange sites for exchanger
e
.
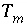 Total quantity of
m
, an element, element valence, exchanger site, surface site, or alkalinity, mol or for alkalinity, eq.
Total quantity of
m
, an element, element valence, exchanger site, surface site, or alkalinity, mol or for alkalinity, eq.
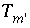 Total quantity of a dissolved element or element valence state excluding alkalinity, hydrogen, oxygen, and electrons, mol.
Total quantity of a dissolved element or element valence state excluding alkalinity, hydrogen, oxygen, and electrons, mol.
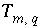 Total moles of an element, element valences, or alkalinity,
m
, in solution
q
, mol or for alkalinity, eq.
Total moles of an element, element valences, or alkalinity,
m
, in solution
q
, mol or for alkalinity, eq.
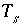 Total number of equivalents of surface sites for surface
s
.
Total number of equivalents of surface sites for surface
s
.
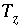 Charge imbalance for the system during reaction and transport calculations, eq.
Charge imbalance for the system during reaction and transport calculations, eq.
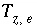 Charge imbalance for the exchanger
e
, eq.
Charge imbalance for the exchanger
e
, eq.
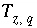 Charge imbalance for the aqueous phase
q
, eq.
Charge imbalance for the aqueous phase
q
, eq.
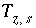 Charge imbalance for the surface
s
, eq.
Charge imbalance for the surface
s
, eq.
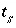 Thickness of diffuse layer for surface
s
, m.
Thickness of diffuse layer for surface
s
, m.
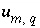 Uncertainty limit assigned to element
m
in solution
q
, mol.
Uncertainty limit assigned to element
m
in solution
q
, mol.
v Pore water flow velocity (m/s).
V Amount of solution in kinetic reactions, kg H 2 O.
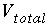 Total volume of a fixed-volume gas phase, L.
Total volume of a fixed-volume gas phase, L.
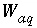 Mass of water in the aqueous phase, excluding any water in diffuse layer of surfaces, kg.
Mass of water in the aqueous phase, excluding any water in diffuse layer of surfaces, kg.
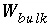 Total mass of water in the system, includes aqueous phase and water in the diffuse layer of surfaces, kg.
Total mass of water in the system, includes aqueous phase and water in the diffuse layer of surfaces, kg.
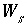 Mass of water in the diffuse layer of surface
s
, kg.
Mass of water in the diffuse layer of surface
s
, kg.
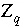 Charge imbalance in solution
q
in inverse modeling, eq.
Charge imbalance in solution
q
in inverse modeling, eq.
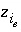 Charge on exchange species
i
e
. (Normally equal to zero).
Charge on exchange species
i
e
. (Normally equal to zero).
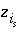 Charge on surface species
i
s
.
Charge on surface species
i
s
.
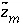 Charge on aqueous master species minus alkalinity assigned to the master species.
Charge on aqueous master species minus alkalinity assigned to the master species.