EXAMPLES
The initial water for this example is defined to be a ground water in equilibrium with calcite at a partial pressure of carbon dioxide of 10-1.5. Pure water is defined with the SOLUTION keyword by using defaults for all values (pH = 7, pe = 4, temperature = 25 C); calcite and carbon dioxide are defined with EQUILIBRIUM_PHASES; and SAVE is used to save the equilibrated solution (table 15). The organic decomposition reaction with a carbon to nitrogen ratio of approximately 15:1 is added irreversibly to this solution in increments ranging from 1 to 1000 mmol (REACTION keyword). A gas phase, which initially has no moles present, is allowed to form if the sum of the partial pressures exceeds 1.1 atm (GAS_PHASE keyword); only CO2, CH4, N2, and NH3 are allowed to occur in the gas phase. SELECTED_OUTPUT is used to print to a file (ex7.pun) the partial pressures and the number of moles in the gas phase of each gas at each step of the reaction.
TITLE Example 7.--Organic decomposition and bubble formation
SOLUTION 1
EQUILIBRIUM_PHASES 1
Calcite
CO2(g) -1.5
SAVE solution 1
SELECTED_OUTPUT
-file ex7.pun
-si CO2(g) CH4(g) N2(g) NH3(g)
-gas CO2(g) CH4(g) N2(g) NH3(g)
END
USE solution 1
GAS_PHASE 1
-pressure 1.1
CO2(g) 0.0
CH4(g) 0.0
N2(g) 0.0
NH3(g) 0.0
REACTION 1
(CH2O)N0.07 1.0
1. 2. 3. 4. 8. 16. 32 64. 125. 250. 500. 1000. mmol
END
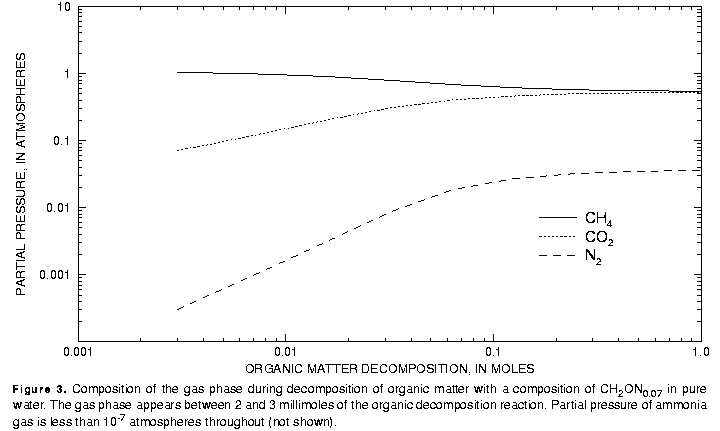
The gas phase appears between 2 and 3 mmol of reaction have been added (fig. 3). Initially the gas is more than 90 percent CH4 and less than 10 percent CO2, with only minor amounts of N2 and NH3 (NH3 partial pressures were less than 10-7 atm throughout the reaction calculation). The volume of gas produced ranges from less than 1 mL at 3 mmol of reaction to more than 20 L after 1 mol of reaction. After 1 mol of reaction is added, nearly all of the carbon and nitrogen is in the gas phase.